Urea; Features and applications

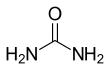
Urea, the chemical compound also known as carbamide, is an organic compound with the chemical formula CO (NH2)2. This amid has two groups of -NH2 which are connected to each other by a carbonyl agent group (C=O).
This solid material is white, odorless, dissolved in water, and almost non-toxic. Carbamide solution is neither acidic nor does it have alkaline properties in water.
Carbamide is used in fertilizers as a nitrogen source (N) and is a critical ingredient for chemical industries.
Friedrich Wohler discovered that CO (NH2)2 could be produced from inorganic ingredients. His theory was a critical climax in chemistry science in 1828. This discovery showed for the first time that material previously used just as a by-product could be synthesized in the laboratory without biological ingredients.
Contents
Urea History
Urea was first discovered in urine in 1727 by a Dutch scientist, ‘Herman Boerhaave’. Although this discovery is attributed to the French chemist, Hillaire Rouelle.
In 1828, Friedrich Wohler, the German chemist, artificially produced carbamide through the treatment of silver cyanate:
AgNCO + NH4Cl → (NH2) 2CO + AgCl
This was the first time an organic compound was synthesized artificially from inorganic ingredients, without the existence of living creatures. This discovery was so important for the development of organic chemistry. This discovery made ‘Wohler’ write enthusiastically to Berzelius: “I should tell you that I can make urea without using human or animals kidney. Ammonium Cyanate is urea.” In fact, this was wrong!
These two distinct chemical substances are similar to N2H4CO in terms of chemical formula which severely follow carbamide in balanced conditions. Despite this, with his discovery, Wohler gained status among organic chemistry ancestors.
Urea production methods

CO(NH2)2 is produced in industrial measure: In 2012, production capacity was about 184 million tons throughout the world.
What is urea made of?
-
Industrial methods for urea production:
For using carbamide in industry, carbonyl diamide is produced from synthesized Ammonia and Carbon dioxide. Due to the fact that great amounts of carbon dioxide are produced in ammonia production procedure as a by-product of hydrocarbons (most frequently natural gas, fewer oil derivations), or sometimes it is made of coal, carbamide production factories are almost always next to this site. The place where Ammonia is produced.
Although natural gas is the most economical and available ingredient for ammonia production, however, powerhouses use that, do not produce carbon dioxide for transforming the total ammonia produced to carbonyl diamide.
In recent years, new technologies have been generated for retaining complement carbon dioxide of combustion exiting gases, produced in the amended furnaces of the Ammonia synthesis gas factory. These plans make ammonia be retained as a separate product and will lead to a reduction in the spread of greenhouse gases.
-
Urea production method through synthesis:
The main procedure developed in 1922, is also called Bosh-Meiser after CO (NH2)2 synthesis procedure discoverers. Different procedures of commercial production of carbamide under conditions this substance is formed are available.
This procedure includes two balanced main reactions with deficit transformation of reactors. The first reaction is the constitution of carbamate: calorific and fast reaction between liquid ammonia and gas carbon dioxide (CO2) in high temperature and pressure for constitution of ammonium carbamate is as follows:
NH3+ CO2⇌ H2N-COONH4
ΔH = -117kJ/mol (in 110 atmospheres and 160 Celsius degrees)
The second stage is the transformation of carbonyl diamide. This analysis procedure is slower and heat sinks. Ammonia carbamate is transferred to urea and water:
H2N-COONH4 ⇌ (NH2) 2CO + H2O
ΔH = +15.5 kJ/mol (in 160-180 Celsius degrees)
The overall transformation of NH3 and CO2 to urea is calorific. The reaction heat of the first reaction is the second procedure factor. Just like all chemical balances, these reactions behave based on Le Chatelier’s principle, and conditions that mostly benefit carbamate formation have an undesirable effect on carbamide transformation balance.
Therefore, the procedure conditions is a “compatibility”: the effect of improper factors in the first reaction with high temperature (about 190 Celsius degrees), compensates with the heat needed for the second stage by performing the procedure under high pressure (140-175 bars) which supports the first reaction.
Although CO2 gas compression is necessary for this pressure, ammonia from refinery in the liquid form is available which could be pumped into the system with more economic efficiency. In order for carbamide to be formed slowly and get balanced, so much atmosphere is needed for the reaction, therefore, a synthesis reactor in a huge urea refinery is a reservoir under pressure.
Due to the fact that this compound transformation is the deficit, the product must be separated from ammonium carbamate without modification. In the carbamide production refinery, this task is performed through pressure reduction of the system to the atmosphere in order for carbamate to be once more analyzed to ammonia and carbon dioxide.
Generally, due to the fact that the recompression of ammonia and carbon dioxide was not economic, at least ammonia is used for the production of other products such as ammonium nitrate or ammonium Sulfate. (carbon dioxide was usually wasted.) Plans presented later could retain ammonia and carbon dioxide.
What are the side reactions in the production of urea in the petrochemical company?
Fortunately, the transformation reaction of CO(NH2)2 is slow. Two successive reactions in this procedure create impurity. Biuret is formed when two carbamide molecules are compounded by losing an ammonia molecule.
2 NH2CONH2 → H2NCONHCONH2 + NH3
What is the undesirable product in urea production?
Normally, this reaction is suppressed through ammonia excessive maintenance in the synthesis reactor. Biuret is so undesirable in urea fertilizer because it is poisonous for arable plants. However, carbamide fertilizer use depends on the product identity and its usage method (when it is used as cow food complement, mixing biuret and carbamide is not an important matter).
Isocyanic acid is made of ammonium cyanide thermal synthesis which is in chemical balance with carbamide:
NH2CONH2 → NH4NCO → HNCO + NH3
In urea production in a petrochemical company, the final product is gained as granule, bullet-shaped, crystalline, and dissolved.
For carbamide’s main application as fertilizer, it is presented to the market in solid form or as a pill or granule. The pill benefit is that overall, they could be produced as cheaper than granules, and this method was completely stabilized in the industrial method a long time before this carbamide granularity procedure was commercialized.
According to the limited size of ingredients produced with a spherical degree, and regarding their low resistance against smash and hit, generally, carbamide solid pill function at the time of saving, displacement, and bulk use is less than granule.
Compound fertilizers with high quality and consisting of nitrogen along with other ingredients such as phosphate are produced from the beginning of the fertilizer industry, however, considering low melting point and urea moisture properties, at the end of the 1970s, three companies began producing fluid bed granularity.

-
Carbamide production method in the laboratory:
Urea could be made of phosgene reaction with primary or secondary amines in the laboratory:
COCl2 +4 RNH2 → (RNH) 2CO +2 RNH3Cl
These reactions continue through an Isocyanate mediator. Through the reaction of primary or secondary amines with isocyanate, asymmetrical carbamides could be achieved. Also, carbamide could be produced by heating ammonium cyanate to 60 Celsius degrees.
NH4OCN → (NH2) 2CO
Urea Properties
-
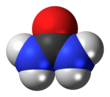
The molecular and crystalline structure
Urea molecule structure is plane. In solid carbamide, the oxygen center is involved in two hydrogen bonds N-H-O. The carbon existing in carbamide is described as a compound of sp2. C-N bonds have the binary bond property and carbonyl oxygen is alkaline compared with formaldehyde. Carbamide’s high solubility in water shows its capability in the extensive bond of hydrogen and water.
Urea has the capability to trap so many organic compounds. In these compounds named clathrates, “guest” organic molecules are formed in channels formed by spirals composed of carbamide molecules connected to hydrogen.
This behavior could be used for mixture dissociations. For example, this property is used in the production of airplane fuel, smoothing oils, and in the separation of hydrocarbons.
-
Carbamide reactions
Carbamide is an alkaline compound. Therefore, it easily gets protonated. Melted carbamide is analyzed to ammonia gas and isocyanic acid.
(H2N)2CO → NH3 + HNCO
Through isocyanic acid, urea is heated and transformed into an extensive spectrum of agglomerated products including biuret, triuret, guanidine, and melamine:
(H2N)2CO + HNCO → H2NCONHCONH2
In water solution, urea is slowly balanced with ammonium cyanate. This hydrolysis produces isocyanic acid which could form carbamylate proteins. Urea reacts with malonic esters and produces barbituric acids.
Urea’s physical and chemical characteristics
| Urea | |
|
|
|
|
|
|
| Compound name | |
| Urea Carbamide |
|
| IUPAC
Urea |
|
| Other names: Carbonyldiamide Carbonyldiamine Diaminomethanal Diaminomethanone |
|
| Identifier Indexes | |
| CAS Number | 57-13-6 |
| E number | E927b |
| PubChem CID | 1176 |
| Properties and characteristics | |
| Urea chemical formula | CH4N2O |
| Molar Mass | 60.056 g·mol−1 |
| Apparent shape | White solid |
| Density | 1.32 g/cm3 |
| Melting Point | 133 to 135 °C (271 to 275 °F; 406 to 408 K) |
| solubility | Glycerol 500 g/L Ethanol 50g/L Acetonitrile ~4 g/L |
| Alkaline dissociation constant (pKb) | 13.9 |
| Thermochemical properties | |
| Formation standard enthalpy (ΔfH⦵298) |
-333.19 kJ/mol |
| Gibbs free energy (ΔfG˚) | -197.15 kJ/mol |
| Flammability point | Inflammable |
| Related compounds | |
| Compounds related to urea structure | Thiourea Hydroxycarbamide |
| Other related compounds | Carbamide peroxide Urea phosphate Acetone Carbonic acid Carbonyl fluoride |
Urea uses
-
Urea uses in agriculture:

More than 90% of carbamide industrial production in the universe has been considered to be used as fertilizer with Nitrogen. Carbamide has the most nitrogen amount among all solid nitrogen fertilizers in usual uses.
The most common impurity of synthesis carbamide is biorite which interferes with plant growth. Carbamide is analyzed in the soil and makes ammonium. Ammonium is absorbed by the plant. In some types of soil, ammonium is oxidized by the bacteria and produces nitrate. This substance, too is among the nutrient substances needed by the plant.
Elimination of nitrogenous compounds in ditches and runoffs is injurious and also harms the environment. Because of this, urea fertilizer is sometimes pre-refined or amended for agricultural uses efficiency increase.
One of the technologies in using carbamide fertilizer is transforming carbamide to derivations such as formaldehyde which is analyzed to ammonia with a speed accordant with plants’ nutritious needs.
-
Urea usage in the provision of resin types:
Urea is the ingredient for the production of several substances’ main groups: urea- formaldehyde and urea- melamine-formaldehyde used in three layered sheet resins.
-
Industrial urea uses in automobile systems:
Carbamide is used for selective non-catalytic reduction reactions (SNCR) and selective catalytic reduction (SCR) in reduction of NOx in contaminants in output gases produced by combustion in diesel engines, bi-fuel and natural gases.
For instance, Blue Tec system injects a carbonyl diamide solution with water base to exhaust system. The ammonia made reacts through hydrolysis of carbamide with nitrogen oxide and is transformed to nitrogen and water in catalytic transformer. Trucks and automobiles using these catalytic transformers should carry diesel output liquids, urea solution in water.
-
Carbamide laboratory uses:
Urea is a powerful protein with 10 m molar densities. This property could be used for an increase in some protein’s solubility. Urea and choline chloride mixture are used as a eutectic solvent (DES), a substance like an ionic liquid. When urea is used in a eutectic solvent, it does not duplicate solved proteins.
Carbonyl diamide can basically act as a hydrogen source for power production in fuel cells. Carbamide existing in drainage could be used directly (however, bacteria rapidly spoil carbamide.)
Carbamide with density of at most 8 m/l could be used in order for brain fixed texture be transparent against light and also, fluorescent signals be kept from cells with label. This provides the possibility of so deeper imaging of neural procedures.
-
Urea uses in medicine:
Cremes with carbamide are used as skin local products for reinforcing skin water supply. Urea is 40% prescribed in psoriasis, dryness, rash, keratosis, keratoderma, callus, and patch.
The blood urea nitrogen (BUN) test is measuring the nitrogen amount in blood derived from carbamide. This substance is used as an indicator of kidney performance. However, compared with other signifier indicators like creatinine, its amount is low. Because this substance level in blood is influenced by other factors such as diet and body water shortage.
-
Urea other applications:
- Carbamide is an element in the output liquid of automobiles with gas oil fuel (DEF) containing 32.5% carbamide and 67.5% nonionized water. For analysis of dangerous spreading gases of NOx to nitrogen and water, DEF is injected to diesel vehicles exhaust stream.
- Carbamide is one of the fodder ingredients and almost cheap source for strengthening animals growth.
- Carbamide is a non-corrosive alternative for salt stone in road defrosting. However, it has lower impact compared with traditional salt or calcium chloride.
- Carbamide is a brown color factor in foods produced in factories.
- Urea is the ingredient in the provision of some skin creams, moisturizers, hair softeners, and shampoos.
- Carbamide along with other salts is counted as a factor in cloud fertilization.
- Carbamide is a flame retardant substance usually used in dry chemical silencers for fire which includes mixture of bicarbonate, urea and potassium.
- Carbamide is an element in many products for tooth whitening.
- Carbonyl diamide along with di ammonium phosphate, as yeast nutrient substance, is used for glucose fermentation to ethanol.
- Carbamide is used as an additive for solubility and moisture conservator in color baths for cloth coloring or printing.
urea in skincare
Urea is a natural moisturizing factor (NMF) that helps the skin retain water and prevent dryness. It also has exfoliating, anti-inflammatory, and anti-microbial properties. Urea can be used to treat various skin conditions, such as eczema, psoriasis, keratosis pilaris, and fungal infections. It is recommended that use products with 5% to 10% urea for daily moisturizing, and products with higher concentrations (up to 40%) for targeted treatments. It is also advised to patch test any new product before using it and to avoid using urea on broken or irritated skin.
read more about how urea benefits the skin, and how to use it in skin care products.
Urea safety
Carbamide compound contact with eye might lead to irritation, redness and pain. This compound might cause skin irritation or redness. It would be harmful if absorbed through skin.
In case of accidental swallow, causes alimentary tract irritation through nausea, vomiting and diarrhea. It may also lead to heart disorders. This could be harmful if completely swallowed.
Inhalation of solid urea powders or solved carbamide vapors might lead to irritation of respiratory tract. Long term or recurrent confrontation to carbamide might lead to negative effects on fertility.
- Chemical consistency: is constant in normal temperature and pressures.
- Adverse conditions for saving and maintenance: bring away from unsuitable substances, cloud production, temperature more than 130 celsius degrees.
- Carbamide incompatibility to other substance: sodium hypochlorite, calcium hypochlorite, sodium nitrate, nitrosyl perchlorate, strong oxidizing factors, dichromates, liquid chlorine, nitrates, permanganate, chromyl chloride.
- Dangerous analytic products: carbon monoxide, nitrogen oxides, carbon dioxide, ammonia.
- Dangerous polymerization: would not happen
Conditions for urea maintenance
Resources or packages containing urea must be kept and saved in storages with suitable ventilation. Minimize production and accumulation of dust made of solid carbamide. Avoid breathing dust, moist and vapors of this substance. Avoid its contact with skin, eye and clothing. Avoid eating food or smoking in storage containing this substance.
Storages and bags containing carbamide must be kept in a dry and cool place. It must be kept in firm containers with closed door.
Urea packing

Urea packing is performed in small bags weighing 50 kg and also in bulk form in 1-ton bags. In loading this product for export from the petrochemical company, loading is usually performed in bulk form. In the standard form, export packings are 50 kg packs.
Packing carbamide fertilizer, just like packing industrial urea packing is performed in 50 kg packs or jumbo bags of 1000 kg and are transferred to the customer considered place with standard trucks or trailers.
Production, market, buying and selling urea status
Urea is produced by state petrochemistries, such as Pardis petrochemical company, Khorasan petrochemical company, Lordegan Petrochemical plant, M.I.S Petrochemical industries, Lavan chemical company, Hormoz petrochemical company, and Kimia Andimeshk petrochemical company. Marketing and exporting industrial urea of state petrochemical companies are such stirring business. As we also mentioned in the above sections, carbamide chemical compound covers different criteria such as agriculture, pharmacy industry, medicine, and laboratory.
Urea fertilizer is an over-consumed product for agriculture section. Urea 46 percent and carbamide 10 percent are produced in granule or prill form in petrochemical company, and according to delivery amount and place, there are different methods for transport. For receiving carbamide price and registration of urea marketing, you can ask Shimico website, industrial chemical substances reference.
Urea Fertilizer Prices Plunge as Gas Costs Ease and Supply Surges
Urea fertilizer prices have tumbled from record levels last year, providing relief to farmers and consumers amid global inflation pressures. Urea, the most widely used nitrogen fertilizer, is now worth about a third of its price in 2022, according to CRU analyst Chris Lawson. The main driver is the decline in natural gas prices, which are used to make ammonia, a precursor to urea. Ample production and exports from Russia and other countries have also boosted supply. Australia, which imports 90% of its urea needs, is building a $6 billion plant in the Pilbara region that will start output by mid-2027. Lower fertilizer costs could help farmers boost yields and lower food prices, which have soared to the highest level since 2011, according to the UN Food and Agriculture Organization.
source: ABC News
Sources:
Last Seen













 Top 10 urea suppliers in Iran
Top 10 urea suppliers in Iran


Users Comment
Sulfur Concrete for Chemical Industries: Definition, Benefits & Uses
Sulfur for Infrastructure: Asphalt, Coatings & Concrete
The Top 10 Industrial Chemicals Shaping the World in 2025
How Urea Can Hydrate and Exfoliate Your Skin
Sodium carbonate| Soda ash; features and applications
Copper Sulfate; Features and applications
Sulfur; Features and applications
Zinc sulfate; Features and applications
Sodium bicarbonate; Features and applications
Ammonium Sulfate; Features and applications
What is the use of caustic soda? An Overview of the caustic soda uses
Caustic soda flakes and the methods of production
16Th International ArabPlast 2023
International Iran Conmine 2023
International Iran Nano 2023
17Th International Iranplast Fair 2023
12th International Composite Expo 2023
International IFarm Agro Fair 2023
22Nd International Paint Resin Coatings Fair
Sodium carbonate| Soda ash; features and applications
Copper Sulfate; Features and applications
Sulfur; Features and applications
Zinc sulfate; Features and applications
Sodium bicarbonate; Features and applications